Abstract
Tularemia is an uncommon but potentially severe zoonotic infection caused by Francisella tularensis, with diverse clinical manifestations that may complicate timely diagnosis. We describe the case of a 79-year-old woman who developed progressive ulcerative hand lesions and systemic symptoms following direct contact with a wild badger while attempting to protect her dog. Her clinical course was marked by delayed recognition, polymicrobial wound cultures, hyponatremia, and profound fatigue, ultimately leading to the diagnosis of ulceroglandular tularemia with systemic features. Diagnosis was confirmed by operative culture and markedly elevated F. tularensis serologic titers. She responded favorably to aminoglycoside-based therapy followed by prolonged doxycycline, with complete resolution of symptoms. This case highlights the importance of detailed exposure history, recognition of atypical presentations, and early consideration of tularemia in patients with nonhealing soft tissue infections and systemic symptoms in endemic regions.
Highlights
- Tularemia is an uncommon but potentially severe zoonotic infection caused by Francisella tularensis with variable clinical manifestations.
- Human infection occurs through diverse exposure routes, including direct contact with infected animals, arthropod vectors, ingestion of contaminated food or water, and inhalation of contaminated aerosols.
- Ulceroglandular tularemia is the most common manifestation and is characterized by an inoculation-site ulcer with tender regional lymphadenopathy that may be accompanied by systemic features.
- Because F. tularensis is fastidious and difficult to isolate in culture, diagnosis is often delayed, particularly when systemic symptoms predominate or early cultures yield nonspecific results.
- F. tularensis is intrinsically resistant to β-lactam antibiotics. Severe or systemic disease is best treated with aminoglycosides, while fluoroquinolones or tetracyclines are options for milder disease or step-down therapy.
1. Introduction
Tularemia is a rare zoonotic infection caused by Francisella tularensis, a highly virulent, facultative intracellular Gram-negative coccobacillus capable of causing severe disease in humans with a low infectious dose [1–3]. Although tularemia occurs sporadically throughout the United States, it remains endemic in several central and south-central states, including Missouri, Kansas, Arkansas, and Oklahoma [1,4,5]. Human infection results from diverse exposure routes, including direct contact with infected animals, arthropod vectors, ingestion of contaminated food or water, and inhalation of contaminated aerosols [1–4].
Clinical manifestations of tularemia are heterogeneous and classically categorized into ulceroglandular, glandular, oculoglandular, oropharyngeal, pneumonic, and typhoidal forms, with considerable overlap depending on the route of inoculation and host factors [1–3]. Delayed diagnosis is common, particularly when presentation is dominated by systemic symptoms or when initial cultures yield nonspecific organisms [2–4]. Older adults and patients with comorbid conditions may be at increased risk for severe or prolonged disease [3,4].
Despite advances in diagnostics and antimicrobial therapy, tularemia continues to pose clinical challenges due to its rarity, protean manifestations, and laboratory handling considerations [1–3,6]. We present a case of ulceroglandular tularemia with systemic features following direct animal exposure in an elderly patient, illustrating the importance of epidemiological context. This report is accompanied by a focused discussion of clinical forms, epidemiology, and current treatment strategies relevant to practicing clinicians.
2. Case Presentation
A 79-year-old woman with a past medical history significant for diabetes mellitus was evaluated for a persistent soft tissue infection following an animal bite.
Approximately one month prior to presentation, the patient heard a commotion near a pond behind her home and found one of her dogs fighting with a badger lodged in a burrow along a creek bed. Unable to scare the animal away, she attempted to protect her dog by grabbing it, sustaining multiple bites and scratches on both hands.
She presented to the emergency department the following day, where the wounds were irrigated, rabies immune globulin was administered, and the first dose of the rabies vaccine series was given. She was prescribed amoxicillin–clavulanate for seven days.
Approximately one week later, she developed worsening erythema and swelling of the hand wounds and was prescribed doxycycline as an outpatient with little improvement.
Several days later, the patient developed profound fatigue, severe headache, and ear pain and returned to the emergency department, where she was found to have hyponatremia with a serum sodium of 126 mmol/L. Hospital admission was recommended, but she declined and ultimately discontinued doxycycline as an outpatient.
Her symptoms continued to progress, and approximately one week later, she returned to the emergency department with worsening fatigue and generalized weakness. She was admitted and found to have multiple bilateral hand abscesses, requiring incision and drainage approximately 18 days after the initial exposure.
Operative cultures initially grew coagulase-negative Staphylococcus and Enterobacter cloacae resistant to amoxicillin–clavulanate. She was treated with piperacillin–tazobactam during hospitalization and was discharged several days later on cephalexin.
When preliminary identification of Francisella species from operative cultures became available shortly thereafter, she was referred for outpatient infectious diseases evaluation.
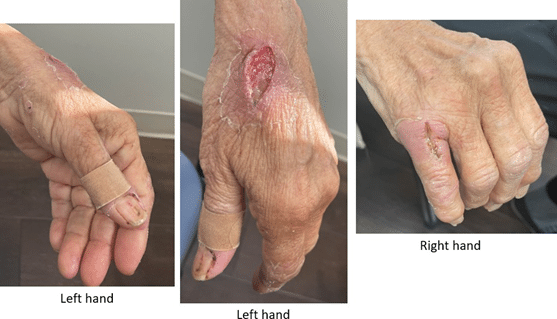
During the outpatient infectious disease evaluation, the patient reported ongoing profound fatigue, anorexia, and weight loss, without fever or recurrent headache. The examination demonstrated healing but inflamed ulcerative and post-incision and drainage lesions involving both hands, as well as localized swelling of the left elbow (Figure 1).

Laboratory studies revealed a white blood cell count of 7.3 ˆ 109/L, hemoglobin level of 10.3 g/dL, platelet count of 330 ˆ 109/L, serum sodium level of 135 mmol/L, creatinine level of 1.06 mg/dL, and normal liver enzymes. Inflammatory markers remained elevated, with an erythrocyte sedimentation rate of 123 mm/h and C-reactive protein level of 3.6 mg/dL.
Serological testing demonstrated a Francisella tularensis antibody titer of 1:1280, confirming tularemia. She was treated with intravenous gentamicin for one week followed by doxycycline for three weeks, with marked clinical improvement at follow-up and complete symptom resolution upon subsequent evaluation.
3. Discussion
3.1. Epidemiology, Reservoirs, and Routes of Exposure
Tularemia is a rare but potentially severe zoonotic infection caused by Francisella tularensis, a highly virulent, facultative intracellular Gram-negative coccobacillus [1–3]. Human infection occurs through multiple exposure routes, including direct contact with infected animals, arthropod vectors, ingestion of contaminated food or water, and inhalation of contaminated aerosols [1–6].
In nature, F. tularensis has been detected in a wide range of wildlife species, including lagomorphs, rodents, carnivores, birds, amphibians, fish, and invertebrates, with lagomorphs and rodents serving as the primary reservoirs [1]. The patient described in this report experienced direct animal contact with a wild badger in a rural setting, representing a classic epidemiological risk factor for tularemia [1–4].
3.2. Clinical Forms, Systemic Overlap, and Severity Considerations
Tularemia classically presents in several clinical syndromes that reflect the route of inoculation and host factors, and these categories often overlap in practice (Table 1) [1–3,5]. The ulceroglandular form is most common and features an inoculation-site ulcer with tender regional lymphadenopathy, typically following animal exposure or tick bite [1–6]. Related syndromes include glandular disease (lymphadenopathy without an evident primary lesion), oculoglandular disease (conjunctival inoculation), and oropharyngeal disease after ingestion of contaminated food or water [1–6]. Pneumonic tularemia can occur after inhalational exposure or via hematogenous pulmonary seeding, while typhoidal tularemia describes a predominantly systemic febrile illness that may resemble sepsis or a prolonged febrile syndrome [2,3]. Importantly, systemic manifestations can accompany localized disease, as in this patient, who exhibited ulceroglandular findings with prominent constitutional symptoms and laboratory abnormalities consistent with overlap toward a typhoidal phenotype [1–6].
| Clinical Form | Key Features |
|---|---|
| Ulceroglandular | Skin ulcer at inoculation site with tender regional lymphadenopathy |
| Glandular | Tender regional lymphadenopathy without visible skin lesion |
| Oculoglandular | Conjunctivitis with regional lymphadenopathy |
| Oropharyngeal | Pharyngitis after ingestion of contaminated food or water |
| Pneumonic | Primary inhalation or secondary hematogenous pulmonary involvement |
| Typhoidal | Systemic febrile illness without localized findings |
Untreated tularemia may lead to prolonged fever, weight loss, lymphadenopathy, and substantial functional decline lasting weeks to months, and relapse has been reported most commonly in ulceroglandular disease treated with doxycycline monotherapy—highlighting the importance of appropriate antimicrobial selection and duration [1–3]. Historical data from the pre-antibiotic era describe mortality approaching 60% in pneumonic and typhoidal tularemia, whereas modern series report overall mortality closer to 2%, reflecting improved recognition and effective therapy [2,4,7]. Systemic features such as profound fatigue, headache, and hyponatremia can increase diagnostic complexity; typical laboratory patterns may include normal or elevated leukocyte counts, elevated inflammatory markers, thrombocytopenia, hyponatremia, elevated hepatic transaminases, and elevated creatine phosphokinase, many of which were seen in this case [1–6].
3.3. Geographic Distribution and Surveillance
Epidemiologically, tularemia remains endemic throughout much of the United States [4–7]. From 2011 through 2022, cases were reported from 47 states, with Arkansas, Missouri, Kansas, and Oklahoma accounting for approximately 50% of reported infections [6]. This geographic distribution aligns with the patients residence in the central United States [1,4,5].
Missouri surveillance data from 2000 to 2007 demonstrated that ulceroglandular and glandular forms predominated among children, whereas adults are more likely to develop pneumonic disease, highlighting age-related differences in clinical presentation [4]. Exposure data from the same region showed that tick bites accounted for the majority of pediatric cases, whereas adults more frequently reported aerosol exposures related to agricultural activities or lawn mowing, although animal exposure remained an important risk factor across age groups [4].
3.4. Diagnostic Challenges and Laboratory Handling Considerations
Delayed diagnosis is common in tularemia, particularly when initial cultures yield nonspecific organisms or when the disease presents with systemic features without classic ulcerative lesions [1–4]. F. tularensis is fastidious and poses a laboratory exposure risk, often leading to delayed or retrospective identification [2–4,6].
In this case, the preliminary growth of Francisella species from operative cultures prompted appropriate serologic testing, which confirmed the diagnosis with a markedly elevated antibody titer [1–3].
3.5. Treatment Considerations and Antimicrobial Selection
Given the rarity of tularemia and the absence of large randomized controlled trials, management relies on observational data, experimental models, and expert consensus. Recommended treatment for tularemia depends on disease severity and clinical form [1–6]. Francisella tularensis is a facultative intracellular Gram-negative coccobacillus with intrinsic resistance to β-lactam antibiotics, including penicillins, cephalosporins, and carbapenems. This resistance is mediated by chromosomally encoded β-lactamases and other intrinsic mechanisms that limit the activity of β-lactam agents against
F. tularensis. As a result, β-lactams are ineffective for tularemia treatment and are associated with delayed clinical response and treatment failure when used empirically [6].
Aminoglycosides, particularly gentamicin and streptomycin, remain the preferred agents for severe, systemic, or complicated tularemia, including pneumonic, typhoidal, and disseminated disease [1,4–6]. These agents exhibit bactericidal activity and favorable intracellular penetration, with streptomycin historically considered the drug of choice and gentamicin widely used due to availability and comparable efficacy [1,4–6]. Gentamicin is not formally approved by the U.S. Food and Drug Administration for tularemia but is strongly recommended by the Centers for Disease Control and Prevention for severe disease, with dosing adjusted for renal function [2,5].
For milder or uncomplicated disease, oral agents such as fluoroquinolones (e.g., ciprofloxacin) or tetracyclines (e.g., doxycycline) are effective alternatives [1–6]. Fluoroquinolones demonstrate excellent bioavailability and intracellular activity and have shown high efficacy in observational studies, whereas doxycycline is associated with higher relapse rates when used as monotherapy, particularly for ulceroglandular disease [1–3,6]. Treatment duration typically ranges from 10 to 21 days depending on agent selection and clinical response [1–6].
Delayed initiation of appropriate therapy is common, particularly when tularemia presents with nonspecific systemic features or polymicrobial wound cultures [1–4,6]. In such cases, empirical regimens often include β-lactam antibiotics, which lack activity against F. tularensis and may contribute to prolonged illness. In the present case, initial treatment with amoxicillin–clavulanate, piperacillin–tazobactam, and cephalexin likely failed to address the underlying infection due to intrinsic resistance [6].
Definitive therapy with intravenous gentamicin followed by prolonged doxycycline treatment resulted in marked clinical improvement and complete resolution of symptoms in our patient, consistent with current treatment recommendations for ulceroglandular tularemia with systemic features [2,5,6].
4. Conclusions
Tularemia is an uncommon but clinically important zoonosis that can present with protean manifestations and significant systemic morbidity, particularly when diagnosis is delayed [1–3].
This case reinforces that tularemia should remain a key consideration in patients with nonhealing skin and soft tissue infections and systemic symptoms after plausible zoonotic exposures in endemic settings. A deliberate focus on exposure history, appropriate diagnostic testing (with laboratory coordination), and prompt initiation of active, non-β-lactam therapy can meaningfully shorten illness duration and reduce the risk of complications, relapse, and prolonged functional decline [1–6].
Funding
This research received no external funding.
Acknowledgments
The author used ChatGPT for content assistance, specifically to create an initial draft from prior personal presented material. The manuscript was reviewed and revised the material generated and takes full responsibility for the content of this publication.
Conflicts of Interest
The author declares no conflict of interest.
References
- Carvalho, C.L.; Lopes de Carvalho, I.; Zé-Zé, L.; Núncio, M.S.; Duarte, E.L. Tularaemia: A challenging zoonosis. Comp. Immunol. Microbiol. Infect. Dis. 2014, 37, 85–96. [CrossRef] [PubMed]
- CDC. About Tularemia. Tularemia. 1 May 2024. Available online: https://www.cdc.gov/tularemia/about/index.html (accessed on 20 February 2026).
- Miller, S.D.; Snyder, M.B.; Kleerekoper, M.; Grossman, C.H. Ulceroglandular Tularemia: A Typical Case of Relapse. Henry Ford Hosp. Med. J. 1989, 37, 73–75. [PubMed]
- Centers for Disease Control and Prevention (CDC). Tularemia-Missouri, 2000–2007. MMWR Morb. Mortal. Wkly. Rep. 2009, 58, 744–748.
- Rich, S.N.; Hinckley, A.F.; Earley, A.; Petersen, J.M.; Mead, P.S.; Kugeler, K.J. Tularemia—United States, 2011–2022. MMWR Morb. Mortal. Wkly. Rep. 2024, 73, 1152–1156. [CrossRef] [PubMed]
- Maurin, M.; Pondérand, L.; Hennebique, A.; Pelloux, I.; Boisset, S.; Caspar, Y. Tularemia treatment: experimental and clinical data. Front Microbiol. 2024, 14, 1348323. [CrossRef] [PubMed]
- Kelson, M.; Burnett, J.; Girgis, S.; Bakr, M. Tularemia: The Resurgence of a Diagnostic Challenge and Clinical Dilemma in the United States. Cureus 2022, 14, e27363. [CrossRef] [PubMed]

